Life comes at you fast.
We’re all affected by the supply chain issues and inflation over the past two years. Few areas have seen as much turmoil as agriculture.
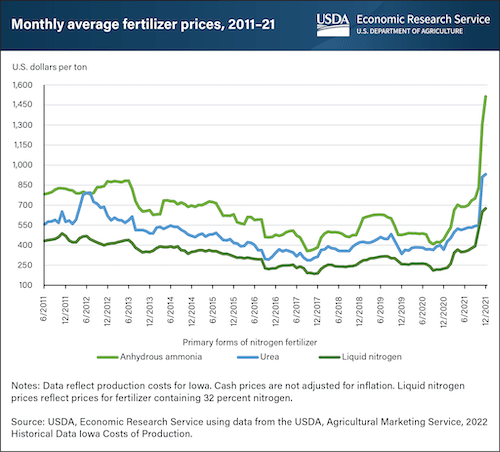
The United States is the largest consumer of nitrogen fertilizers, importing 14.5% of the world’s supply. The cost of nitrogen-based fertilizers has quadrupled since 2020. To make matters worse, Russia, the world’s largest exporter of synthetic fertilizers, banned exportation of fertilizers in early 2022.
Oh, and they banned exports before they invaded Ukraine, which further isolated Russia from global trade.
Conventional farmers who rely on synthetic fertilizers are struggling with high costs and low returns. Commodity crop growers depend on subsidies from the government because of extremely low profit margins. The skyrocketing costs of fertilizer and fuel may be the tipping point that gets farmers to consider another means of crop fertilization.
This article will summarize the history of synthetic fertilizers and their very real costs to the environment.
We’ll also stress that these chemicals are not needed in the first place and we’ll go on to discuss other natural sources of the nitrogen essential for growth.
The History of Synthetic Nitrogen
It’s useful to look at how synthetic fertilizer, specifically anhydrous ammonia got its start.
In the late 1800s and early 1900s, German scientist Fritz Haber and his team converted atmospheric nitrogen into ammonia through a high-pressure, high-temperature procedure, which came to be known as the Fritz-Haber Process.
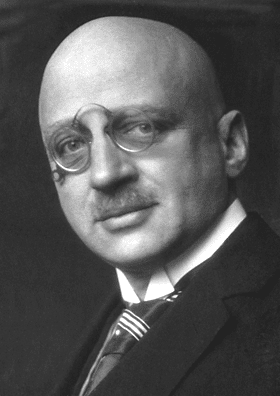
Through a partnership with Carl Bosch at BASF, fertilizer production was scaled to industrial levels. The increased plant yield from the Haber-Bosch Process prevented mass starvation, earning Haber the Nobel Prize in Chemistry in 1918.
Unfortunately, Haber was also instrumental in developing German chemical weapons – and defended their use – sullying his reputation today.
The overlap between synthetic fertilizer and munitions manufacturing was so great that factories dedicated to one could be used for the other.
So post-World War II munitions factories – in Germany and beyond – began retooling for post-war fertilizer production.
Today, literally half of the world’s food production is dependent in some way on Haber-Bosch.
While fertilizer proponents can point to food production as a good for the world, synthetic fertilizer destroys soil biology.
We now know plenty of ways for nutrients to be cycled within an ecosystem without the need for chemical fertilizers. As growers and land stewards, our number one job is to make sure that this soil life is present to work with the plants we are trying to grow.
The True Cost of Synthetic Fertilizers

The Economic Cost of Synthetic Fertilizers
Farmers who rely on petrochemical fertilizers are feeling the burn more than ever in 2022.
In 2020, the price of anhydrous ammonia – the most common form of conventional nitrogen – was around $450 per ton.
As of January 2022, these prices were nearing $1500 per ton.
The future of conventional farming is uncertain. Many farmers don’t even know if they can risk putting any seeds in the ground with the higher prices for chemicals and fuel.
The Environmental Cost of Synthetic Fertilizers
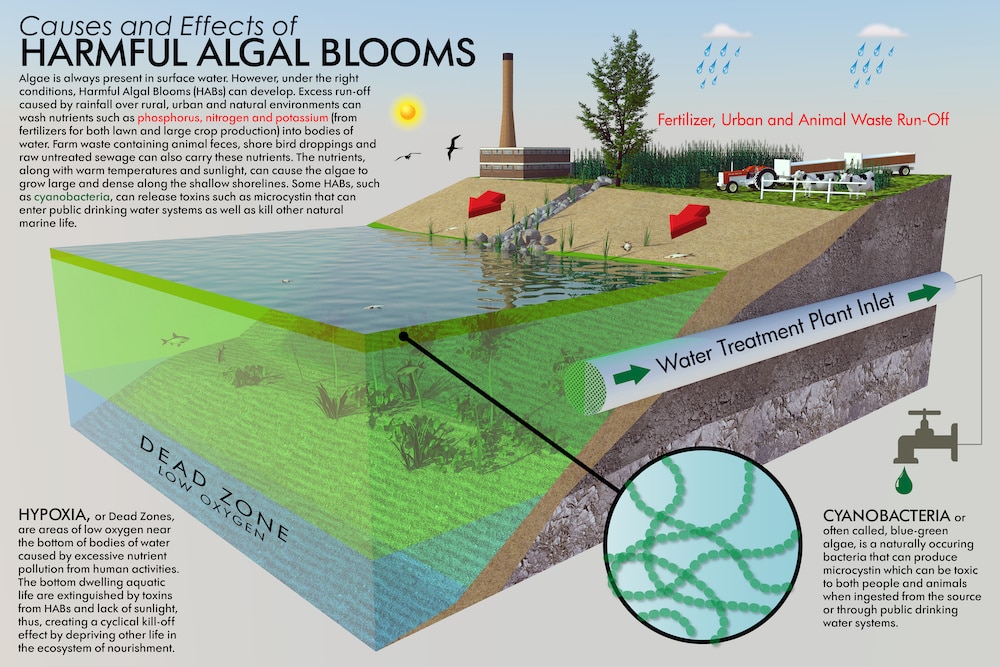
Plants take up only a small percentage of these chemicals, most of which seep down into the water table and then move via stream or river to larger bodies of water. Every year, the Gulf of Mexico and the Great Lakes in the United States experience vast algal blooms. These blooms are the result of large amounts of fertilizers entering waterways and eventually making their way to oceans and lakes.
Algal blooms use up a majority of the oxygen in these bodies of water causing large amounts of aquatic species to die off. That extra nitrogen fertilizer that a farmer in Illinois applies to their corn doesn’t just stay in the soil. It can travel 1000 miles to the Gulf.
Algal blooms occur all over the United States and are the cause of potentially toxic algae in ponds, rivers, lakes, bays, and drinking water supplies. A 2020 analysis estimated in the past decade the U.S. has spent over a billion dollars to deal with these outbreaks of algae.
What if none of this was necessary?
What if we chose to use natural methods of fertility for growing food crops and landscapes?
What if the Earth and its atmosphere held all the nutrients and minerals that plants need?
I’ll tell you a secret. . . . . It already does.

Getting Away From Chemical Fertilizers: What Do We Do Now?
For millions of years, plants and microorganisms worked symbiotically before humans were ever on the planet.
Plants can manage their own nutritional requirements.
Plants use photosynthesis to convert energy from the sun into chemical energy that is stored or used as carbohydrates. The plant exudes some of these carbohydrates- mainly sugars, starches, and proteins – through their roots to attract bacteria and fungi.
These microbes have excess nutrients stored within them, so when they are consumed or die, they release the nutrients in a plant-available form. Since the plant has attracted the bacteria and fungi to the root zone, the nutrients from them are readily taken up and used by the plant.
How do we replace this life in the soil?
How can growers provide crops with the nitrogen essential to their growth?
The number one way to inoculate soils (and soilless mediums) with beneficial biology is through quality composts, vermicomposts, and liquid compost amendments like teas and extracts.

Vermicomposts are especially known for their higher populations of microorganisms and plant growth hormones making them the number one choice for adding biology to soils and soilless mediums.
Vermicompost has another added advantage.
Worms have a calciferous gland that excretes calcium carbonate through their digestive system, resulting in higher quantities of calcium in worm castings, a required mineral for nitrogen uptake in plants.
Vermicompost can also be made into a compost tea or extract.
Compost teas and extracts are much more efficient than granular compost at getting coverage onto plants and soils. They can also be applied to the foliage of plants providing a protective layer of beneficial biology which helps to shield against pests and disease.
Organic Sources of Nitrogen
Organic nitrogen is delivered in various forms.
Manure-based forms are the most readily used option on medium to large-scale farms due to the cost and availability. Everything poops, right?
Poultry manure has one of the highest concentrations of nitrogen per unit. Most manures will need to be aged or composted in order to not “burn” plants due to the excessive nitrogen. Other types of manure used for fertilizers come from the rear ends of sheep, pigs, cattle, rabbit, alpaca, and llama.
Thermophilic composting above 131°F kills pathogens or weed seeds present in manure. But composting also works to dissipate the manure within all of the other organic material in a compost pile, helping to spread the nitrogen over a greater area.
A word of caution when it comes to manures…..

Be leery of grazers and ruminants who possibly have access to ditches or fields sprayed with chemical herbicides. Many horses and cattle browse on grasses or straw sprayed with persistent herbicides.
Persistent herbicides can remain active for close to two years, even after digestion by animals. They may end up in composts where the organic matter covered in herbicides is broken down into even smaller particle sizes, making it much more spreadable. Composts containing these persistent herbicides can kill or deform future crops. It only takes one part per billion (the equivalent of one drop in an Olympic-sized pool) to have an effect on plants.

So if the environmental or financial costs of synthetic fertilizer are driving us away from synthetic nitrogen, what are we to do?
What are some natural sources of nitrogen we can use, especially at smaller scale?
Larger farmers will likely want local nitrogen sources due to shipping cost, but at small scale, the following organic nitrogen products will help you to replace any dependence you might have on synthetic fertilizer.
Animal-Based Nitrogen
Compared to the plant-based nitrogen sources mentioned below, animal-based nitrogen sources tend to be higher in nitrogen.
Feather Meal
A byproduct of the poultry industry, feather meal contains the highest forms of N of any organic fertilizers. Feathers go through a process of grinding and drying until they become a grainy powder.
Average N content: 13-14%
Pros: Slow-release form of nitrogen. Highest form of N. Low cost.
Cons: Not very soluble in water
Blood Meal
Blood meal fertilizer mainly comes from slaughtered animals, particularly cattle. The blood is collected, dried, and turned into a powder.
Average N content: 12-13%
Pros: The most bio-accessible form of high-nitrogen fertilizer. Low cost.
Cons: Easy to use too much. Does not last long. May attract carnivorous animals from nearby.
Fish Meal
The majority of fish meal comes as a byproduct of the food fish and fish oil industry. It is made from the inedible portions of larger fish or from smaller fish who have had the oil expelled.
Average N content: 8-10%
Pros: Great source of phosphorus and potassium as well as micronutrients
Cons: Smells like fish! This is potent stuff.
Crab and Shrimp Meal
Both crab and shrimp meals are byproducts of the seafood industry. The shells and exoskeletons of these crustaceans are dried and ground into a powder form.
Average N content: 4%
Pros: High in calcium, magnesium, and trace minerals. Great for promoting soil biology. More of a “slow-release” form of N. The only meal that features chitin.
Cons: Could emit odors attractive to animals.
Human Urine

There is one other “easy pee-zy” form of nitrogen fertilizer becoming popular (some would even say its #1)…….human urine.
Also known as “peecycling”, human urine is a largely-overlooked source of nitrogen for crops. Rich Earth Institute collects and reclaims urine from local participants and uses it as fertility for croplands. When it comes to using human urine as a source of nitrogen for crop growth, we say “go” for it!
We’d put a check prices button here, but instead, we’ll just ask you to wait until your bladder is full!

Plant-Based Nitrogen
Plant-based nitrogen fertilizers contain lower percentages of nitrogen, demanding a higher rate of application.
Like animal-based fertilizer, plant-based fertilizers can be sprinkled around the root zone or added to potting mixes for seed sowing.
Alfalfa Meal
Alfalfa meal comes from the high-nitrogen alfalfa plant . Alfalfa is dehydrated and ground into a powder or pellet form.
Average N content: 3%
Pros: Sustainable source of nitrogen. Can be grown on-farm. Too much will not burn plants.
Cons: Pelleted forms need rain or moisture to expand and start releasing N.
Soy Meal
Most soy meal is a byproduct from making soybean oil. Soybeans are crushed and the oil is extracted through pressing or the use of solvents. The remnants from this process are then ground to make a meal. Soybean meals may or may not contain soybean hulls.
Average N content: 7%
Pros: One of the highest forms of plant-based N. Slow-release fertilizer.
Cons: Occasionally burns plants or can cause reduced germination. Higher applications of soybean meal brings higher levels of soluble salts.
Cottonseed Meal

Cottonseed meal comes about from the process of extracting cottonseed oil. The process is the same as the extraction of soybean oil, either by pressing or through solvents.
Average N content: 6-7%
Pros: High percentage and slow-release form of N
Cons: Can make soil acidic.
Biology Makes It Happen
I had an ideal learning experience while spraying compost tea for a client. I checked in with the client a few days after applying compost tea to her raised bed vegetable and flower gardens. She noted that “the compost tea burned her tomato plants”.

I offered my apologies but politely explained that the compost tea is made up of 20 gallons of water with about 10 cups of compost plus a few tablespoons of humic acid and fish hydrolysate. This is all diluted in 100 gallons of water. It is impossible for that amount of materials diluted in 120 gallons of water to have a burning effect on a plant.
I asked whether she had applied anything to the plants or the soil before my application of the compost tea. She explained that she had sprinkled a healthy amount of blood meal around the base of her tomato plants because she read that it would help ward off ants. It had mixed in with the color of the soil and so I hadn’t noticed anything out of the ordinary.
All of the bustling microbial life in the compost tea had activated the blood meal and cycled all of that nitrogen extremely fast to the tomato plants effectively “burning” them.
This just goes to show that microorganisms in compost tea work with plants and provide nutrients that are available in the soil.
Just not so much next time!!
Microbes: Literally Pulling Nitrogen From Thin Air
The Earth’s atmosphere is 78% nitrogen. In nature, nitrogen-fixing bacteria pull this nitrogen gas out of the atmosphere and convert it to a plant-available form in the soil, literally creating plant-available nitrogen out of thin air. The Haber-Bosch Process mentioned above is a man-made ( and more energy-intensive!) way to accomplish the same thing.
Some of these bacteria are free living in the soil and some are symbionts, or mutualists with host plants.
Free living forms of nitrogen-fixing bacteria in the soil include Azotobacter, Clostridium, and cyanobacteria (aka blue-green algae). Examples of mutualistic species are Rhizobium which form associations with leguminous plants, Frankia which works symbiotically with dicotyledonous plants, and Azospirillum which associate with cereal grasses.
These mutualistic nitrogen-fixing bacteria penetrate the root hairs of their hosts and form root nodules where both bacteria and plant cells multiply.
Inside the root nodules, the bacteria convert free nitrogen from the atmosphere into ammonia that the host plant uses for growth.
Certain nitrogen-fixing bacteria have even shown to work simultaneously with arbuscular mycorrhizal fungi to amplify nitrogen fixation by increasing the size and number of these root nodules.
You can purchase individual species of nitrogen-fixing bacteria in order to inoculate seeds before planting. Nitrogen-fixing cover crops can grow alongside cash crops during the growing season, or in place of cash crops in the off-season. Nitrogen-fixing cover crops planted in rotation before a high-nitrogen demand crop can provide the soil with the needed nitrogen to allow that cash crop to flourish, negating the need for chemicals whatsoever.
Cover crops also provide living roots to feed soil biology, helping to keep nutrients in the soil and cycle them as needed. By choosing a mix of cover crops with varying root depths, plants can extract an even greater variety of nutrients and minerals from the earth. After these cover crops are terminated or die, they will decompose, making various nutrients and minerals available through microbial interactions.
Kudos To Mother Nature
When it comes to keeping plants and soils healthy, it seems nature knows good and well how to take care of itself. If humans will choose to promote life, instead of fighting it.
The high and hidden costs of chemical fertilizers can be a thing of the past. Biological farming is cost-saving, profit-increasing, soil-building, environment-enhancing….Shall I go on?
By using small amounts of natural forms of organic fertilizers alongside active biology in vermicomposts, composts, and liquid compost amendments plants can obtain all of the nutrients they need. Farmers and gardeners can assist plants and soil microbes in harvesting sun and atmospheric energy to then convert into chemical energy for plant growth and health.
Keeping plants in the ground in all seasons through the use of cover crops feeds the soil as well as the microorganisms, therefore feeding future crops. It is the life in the soil that makes all of this possible.
It seems that maybe we could ALL learn a lesson from soil biology.
Teamwork truly does make the dream work.

If you want to know if your soil is appropriate for your crop, or if you need help our help with anything else, engage our services today!











Thank you, very good information. I would love a print button on your article’s.
I am a current student of Dr Elaine’s The Soil Food Web. LOVE this article. Love that you hired Troy. Get the word out and let’s stop destroying our planet with chemical agriculture!
Me too, the poop loop is the key
Greetings Troy,
Thank you for the informative post! I’ve been wanting to learn more about the Haber-Bosch Process and in particular the ways that “half of the world’s food production is dependent in some way on Haber-Bosch.” Could you point me in the right direction for following up on the numbers? Are there any particular sources you would recommend?
Thank you!
William
Check this one William! One of a few different places I’ve seen the stat.
Cheers!
Steve
Fritz Haber received the Nobel prize in Chemistry in 1918 (not 2018). Additional sources of Nitrogen: Shrimp Meal: 6-6-0. Neem Seed Meal: 6-1-2. Bat Guano: 7-3-1.
While the idea of weaning people away from the use of chemical fertilizers to more natural sources might be admirable, I believe that now is not the time. What with the world in crisis, gas prices climbing steadily, Ukraine in shambles and Russia shutting off all exports to the West in response to sanctions against it, I feel like these times more resemble the days leading up to the Stock market crash of 1929. Then; one day things were bright and wonderful and the next day people were converging on the banks to try and retrieve their savings before the cash ran out. Consider this scenario. For years you’ve had a good relationship with farmer Brown, easily purchasing quantities of his cow manure at a fair price, not storing away a couple extra tons (against a time of emergency) because supply always outstripped demand. Now suddenly there’s a global crisis and chemical fertilizers are in short supply. Dozens of people are converging on farmer Brown looking to buy his cow manure and offer to pay far more than what you paid in the past. One person with much deeper pockets than yours, starts a bidding war and before you know it the price is at 10 times what you can afford; and so you end up with nothing. The motto of the Boy Scouts was: “Be Prepared” and in commercial air travel they say to adjust your own oxygen mask, before trying to help others. In prepping there’s the Prepper Mindset. There needs to be something similar here. In ages past before the advent of big-box stores, people had large pantries and root cellars. Nowadays with the ease of large supermarkets, most people don’t store anything. If you have the ability financially to put away a couple extra 50 lb. sacks of fertilizers and soil amendments, now might be a good time to do that. Caveat Emptor.
Hi Shaul,
Unless Troy fixed it after your comment, I think we got the dates right. I’ll be honest that I’m not sure what solution you’re suggesting here. I don’t think the battleship gets turned around overnight but if not in the middle of a fertilizer price spike, when would we change? I think stockpiling some fertilizer now is probably a good idea, but Farmer Brown hasn’t started moving to organic by now, what’s he going to do if prices get to 10x instead of “only” 5x?
Troy,
Being a novice soil nerd, I enjoyed your article. I am amazed big Ag hasn’t transitioned to organic methods prior to now. The cost of inputs is drastically lower (each year) and the benefits to the environment are enormous, not to mention a healthier product. I have a 1K sq ft sandy field that I have been organically soil building x 4 yrs. This will be my first to grow edible produce. Can’t wait to compare garden vs grocery store brix levels!
Awesome Tonda!
I’m Very greatful about your article how about healthy root fertilizers get you give me some insight